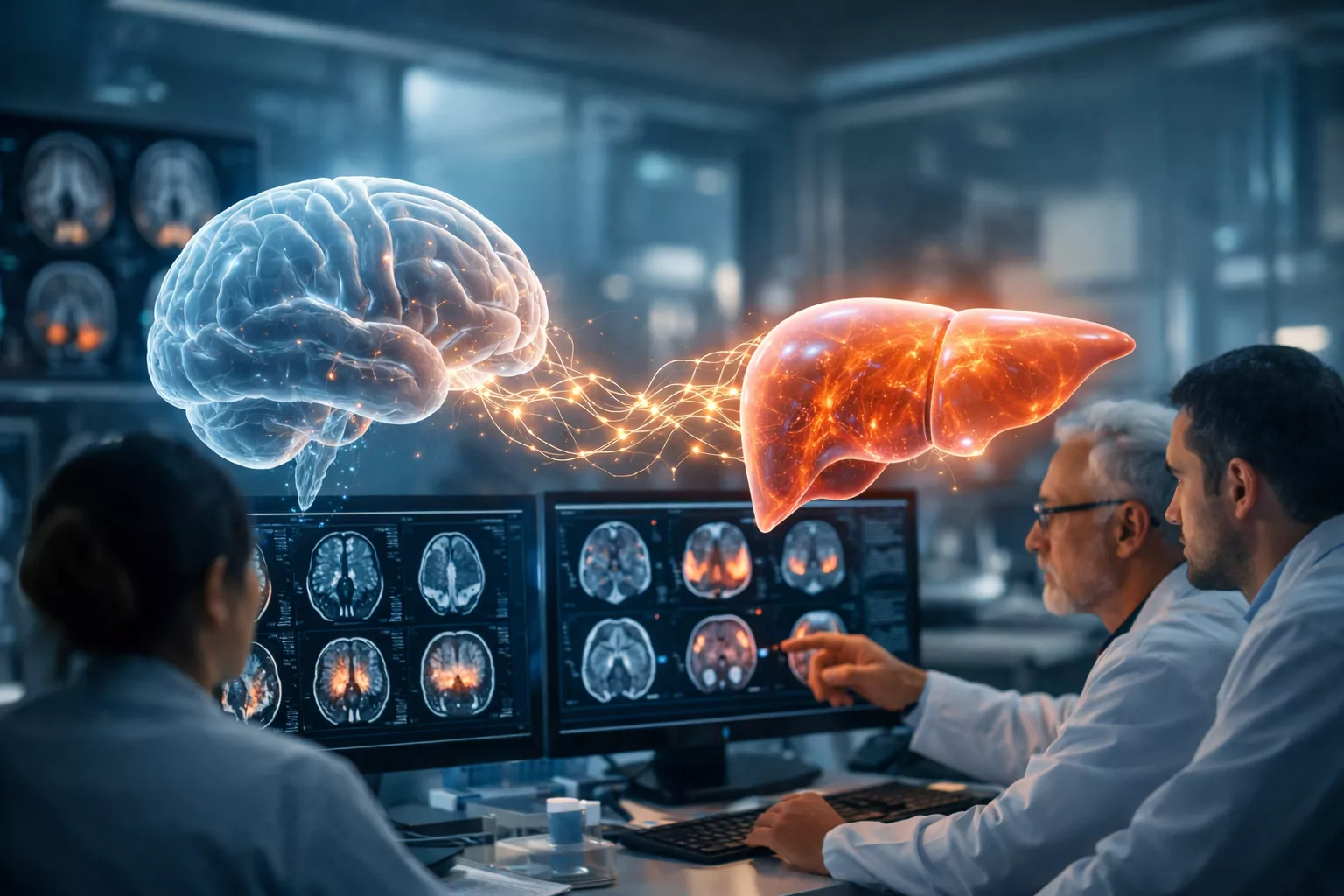
Researchers at the Feinstein Institutes identify noninvasive biomarkers that detect early brain changes in acute liver injury, enabling faster intervention and improved patient outcomes.
Scientists at the Feinstein Institutes for Medical Research, the research arm of Northwell Health, have unveiled a promising noninvasive method to detect early and potentially reversible brain changes associated with acute liver injury (ALI) and its most severe form, acute liver failure (ALF). Their findings offer new hope for improving diagnosis and treatment of hepatic encephalopathy—a serious and often difficult-to-diagnose neurological complication that can arise when the liver rapidly loses its ability to function.
Acute liver injury is a sudden and severe deterioration of liver function, frequently triggered by drug toxicity such as acetaminophen overdose, viral infections, or other toxic insults. While the liver is the primary organ affected, the consequences extend far beyond it. As liver function declines, toxins that are normally cleared from the bloodstream accumulate and can cross into the brain, leading to a cascade of neurological disturbances. In its advanced stages, this can result in hepatic encephalopathy, characterized by confusion, brain fog, altered consciousness, and in severe cases, coma and death.
Despite the grave risks, early detection of brain involvement in ALI and ALF has remained a major clinical challenge. Symptoms of hepatic encephalopathy can be subtle in the beginning, and standard diagnostic tools often fail to capture the earliest biological changes occurring in the brain. Recognizing this critical gap, researchers at the Feinstein Institutes set out to identify objective, measurable brain signatures that emerge before irreversible neurological damage sets in.
The study was first-authored by Santhoshi Palandira, MD, PhD, and led by Valentin A. Pavlov, PhD, professor in the Institute of Bioelectronic Medicine at the Feinstein Institutes. Their findings were published in the peer-reviewed journal Theranostics. The research team employed an advanced imaging strategy using noninvasive dual radiotracer microPET brain scans in a mouse model of acetaminophen-induced acute liver injury that progresses to acute liver failure.
MicroPET imaging is a highly sensitive form of positron emission tomography tailored for small-animal research. By using two radiotracers simultaneously, the researchers were able to capture multiple biological processes in the brain at once. Specifically, they measured changes in brain energy metabolism and neuroinflammation—two interrelated processes that play a critical role in the brain’s response to systemic injury.
One of the most significant discoveries was the identification of what the researchers describe as “neuroinflammetabolic” signatures. These are overlapping hotspots in the brain where both inflammation and altered energy metabolism occur simultaneously. Such changes reflect a stressed and dysregulated neural environment. Importantly, these hallmark changes were detected as early as 24 to 48 hours after the onset of acute liver injury—well before overt neurological symptoms would typically be recognized in a clinical setting.
The affected brain regions included several critical structures responsible for cognition, emotion, autonomic regulation, and coordination. Among them were the thalamus, which serves as a central relay station for sensory and motor signals; the hippocampus, crucial for memory formation and spatial navigation; the hypothalamus, which regulates hormonal and metabolic balance; and the cerebellum, essential for motor control and coordination. The identification of neuroinflammetabolic hotspots in these regions underscores the widespread and multifaceted impact of acute liver injury on brain function.
Beyond localized inflammation and metabolic disruption, the researchers also examined brain network connectivity. By analyzing metabolic connectivity patterns, they discovered distinct alterations in how different brain regions communicate with one another. Some neural networks showed increased connectivity, while others demonstrated significant losses. This pattern of both gains and disruptions suggests that acute liver injury profoundly reshapes the brain’s overall functional architecture.
Such connectivity changes are particularly important because the brain operates as an integrated network rather than as isolated regions. When communication pathways are altered, even subtle changes can translate into noticeable cognitive and behavioral impairments. The ability to detect these network-level disruptions at an early stage offers a powerful tool for predicting disease progression and evaluating therapeutic interventions.
Dr. Pavlov described the platform as a groundbreaking step forward in brain-liver research. According to him, the ability to noninvasively detect characteristic neuroinflammetabolic signatures during the early stages of acute liver injury provides a crucial window for intervention. By identifying patients at risk of developing hepatic encephalopathy before severe symptoms emerge, clinicians may be able to initiate targeted therapies sooner, potentially preventing irreversible brain damage.
This approach also aligns with the principles of precision medicine. Rather than relying solely on clinical symptoms or generalized laboratory markers, physicians could use objective imaging biomarkers to tailor treatment strategies to an individual’s specific neurobiological profile. Additionally, the same imaging platform could be used to monitor the effectiveness of therapies in real time, ensuring that interventions are achieving the desired impact on brain inflammation and metabolism.
The implications of the study extend beyond liver disease. The researchers noted that their novel imaging strategy could be applied to other disorders characterized by peripheral immune and metabolic dysregulation. For example, sepsis-associated encephalopathy—a condition in which systemic infection leads to brain dysfunction—shares similar inflammatory and metabolic pathways. By adapting this platform to different disease contexts, scientists may gain new insights into how systemic illnesses affect the brain.
The study was the result of a collaborative effort involving multiple experts across disciplines. Dr. Pavlov worked closely with David Eidelberg, MD, professor in the Institute of Molecular Medicine; An Vo, PhD, associate professor in the same institute; Yilong Ma, PhD, professor in the Institute of Molecular Medicine; Joseph Carrion, PhD, director of the microPET laboratory; and several other scientists. This multidisciplinary team combined expertise in neuroimaging, immunology, molecular medicine, and bioelectronic medicine to advance understanding of brain-body interactions.
Kevin J. Tracey, MD, president and CEO of the Feinstein Institutes and Karches Family Distinguished Chair in Medical Research, emphasized the broader significance of the findings. He noted that studying the brain’s complex response to acute liver injury reflects the institution’s mission to generate knowledge that leads to cures. By uncovering how systemic organ failure influences neural networks, the research opens new pathways for understanding and treating diseases that bridge the brain and the body.
Ultimately, this work represents a significant step toward closing a long-standing diagnostic gap in acute liver disease. Early identification of brain involvement could transform the clinical management of patients with ALI and ALF, shifting care from reactive treatment of advanced symptoms to proactive, targeted intervention. As research progresses and similar imaging techniques are translated into clinical practice, patients facing life-threatening liver conditions may benefit from earlier diagnosis, improved monitoring, and more personalized therapies—offering renewed hope in a field where rapid deterioration has often left little time to act.